GC-MS vs MRR: A Faster, More Specific Workflow for Chemical Analysis
Gas Chromatography-Mass Spectrometry (GC-MS) remains a standard analytical workflow for volatile and semi-volatile compounds, but it is not always the simplest path for targeted testing. In excipient testing and essential oil analysis, GC-MS workflows can require chromatography optimization, matrix-specific method development, and additional effort to distinguish closely related compounds with high confidence. Molecular Rotational Resonance (MRR) spectroscopy offers a different approach: direct, molecule-specific identification based on each analyte's unique rotational spectrum. For targeted applications such as excipient impurity screening and essential oil authenticity, MRR can simplify workflows while improving specificity for structurally similar compounds.
BrightSpec application notes show how MRR can reduce chromatography-dependent method development, simplify sample handling in challenging matrices, and provide structure-specific confirmation in applications where GC-MS workflows often become more complex. This article compares GC-MS and MRR across three real-world use cases: ethylene glycol and diethylene glycol testing in PEG-400, ethylene oxide and acetaldehyde testing in PEG-3350, and stereoisomer differentiation in essential oil and natural product analysis.
What is the difference between GC-MS and MRR?
With GC-MS, chemists separate compounds chromatographically and then conduct mass analysis, while MRR identifies target compounds by their unique rotational spectra. In targeted workflows, MRR can reduce chromatography-dependent method development, simplify interpretation of structurally similar compounds, and provide a highly specific alternative or complement to GC-MS.
- GC-MS: Separation-first workflow that depends on chromatographic behavior, retention, and mass spectral interpretation.
- MRR: Structure-specific workflow that measures gas-phase rotational spectra tied directly to molecular geometry.
- Best fit for MRR: Targeted testing where specificity, speed, or simplified setup matter more than broad untargeted screening.
Why do GC-MS workflows become complex in targeted analysis?
GC-MS is powerful and well established, but targeted workflows can become time-intensive when compounds are difficult to separate chromatographically or when structurally similar analytes must be distinguished with high confidence. In practice, teams may need to optimize chromatographic conditions, temperature programs, sample preparation, and matrix-specific methods before routine testing is ready. These challenges become more noticeable in excipient testing and natural product analysis, where low-level contaminants or closely related stereoisomers can complicate interpretation.
- Chromatography-dependent method development: GC-MS often requires retention-focused optimization and method tuning.
- Matrix-specific adjustments: Viscous pharmaceutical excipients can require dilution, filtration, or injection-focused handling.
- Isomer specificity challenges: Similar compounds may require stronger separation or additional confirmatory steps.
- Operational overhead: Multi-step GC-MS workflows can add time and complexity before routine testing is established.
How does MRR simplify targeted workflows compared with GC-MS?
MRR simplifies targeted workflows by replacing chromatography-dependent separation with direct, structure-specific detection of known analytes. Instead of relying on retention behavior alone, MRR confirms compounds through their unique rotational fingerprints. In the right applications, this can reduce method development burden, minimize manual preparation steps, and shorten the path from sample to result.
- Target compounds are selected up front. MRR is optimized for known analytes rather than broad untargeted screening.
- Spectral fingerprints drive identification. Each analyte is confirmed by a unique rotational spectrum rather than only retention and fragmentation behavior.
- Chromatographic separation is not required for these targeted assays. This simplifies routine execution and reduces dependence on retention-based method optimization.
- Automation supports throughput. BrightSpec application notes highlight autosampler-based workflows and batch-ready method setup.
How does MRR compare to GC-MS for excipient testing?
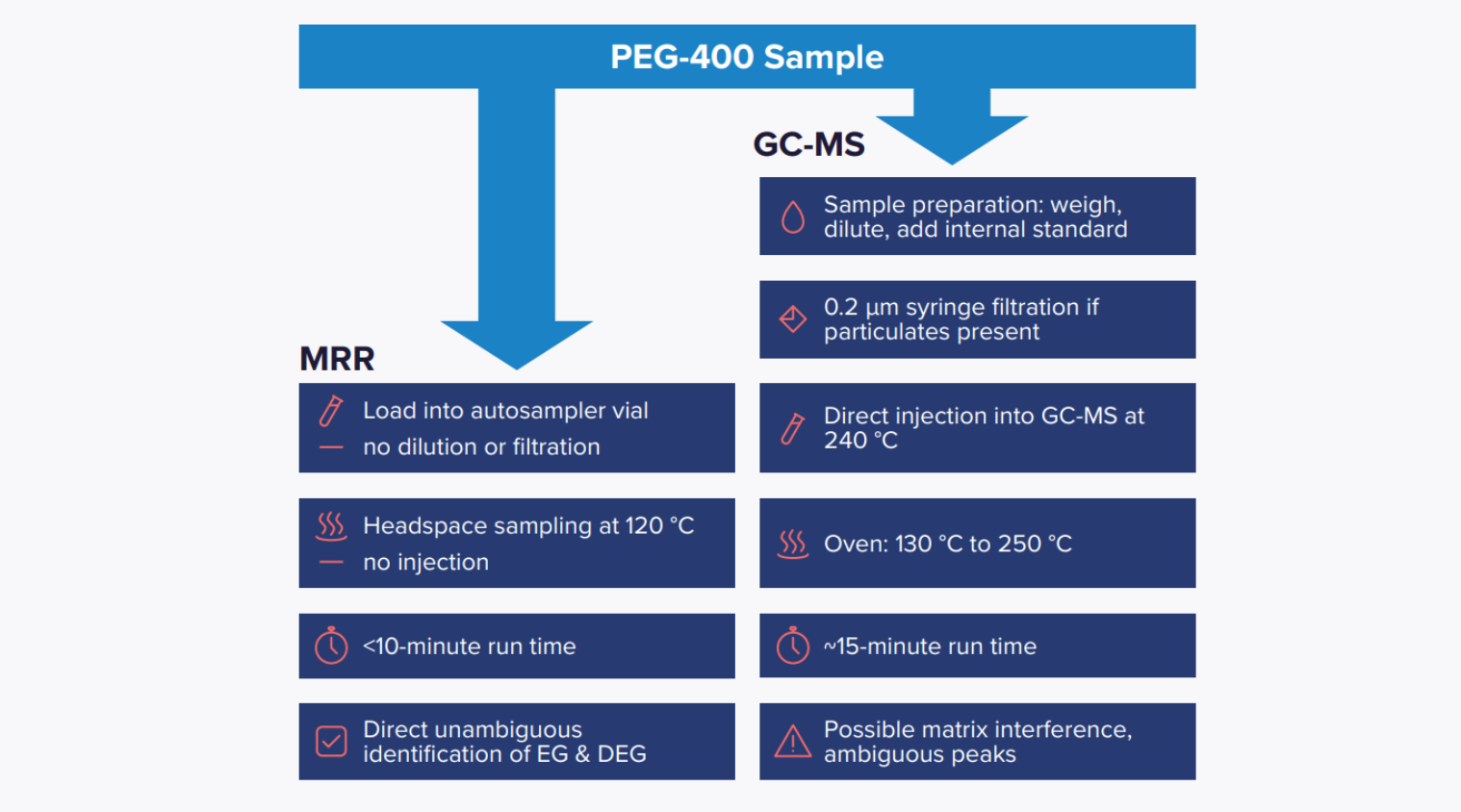
For pharmaceutical excipient testing, the workflow difference is often operational. Headspace GC-MS methods can require matrix-specific optimization and chromatography-focused development before routine analysis. In BrightSpec's excipient testing application notes, MRR is positioned as a targeted alternative for compounds such as ethylene glycol, diethylene glycol, ethylene oxide, and acetaldehyde. By removing the need for chromatographic separation in these targeted analyses, MRR can simplify method development and streamline testing for known analytes in challenging excipient matrices.
PEG-3350 and PEG-400 are both common formulations of Polyethylene Glycol, but they have very different physical properties and purposes. PEG-3350 is typically used in laxative products, and PEG-400 is typically used as a solvent in topical pharmaceutical formulations. Both are highly viscous which presents injection challenges for standard GC-MS workflows.
How does MRR help with EG and DEG testing in PEG-400?
In the PEG-400 application note, MRR is used to quantify ethylene glycol (EG) and diethylene glycol (DEG) through headspace analysis rather than a traditional injection-heavy GC-MS workflow. The application note shows a simpler sample path, with PEG-400 loaded directly into headspace vials and analyzed without the dilution and filtration steps commonly associated with GC-MS workflows in viscous matrices.
BrightSpec reports that EG in PEG-400 achieved strong linearity (R2 = 0.9967) across 10-2960 ppm, with a 3 ppm LOD, 10 ppm LOQ, repeatability of 3.02%, and a 200-second measurement time. DEG in PEG-400 achieved R2 = 0.9868 across 182-2973 ppm, with a 60 ppm LOD, 182 ppm LOQ, repeatability of 3.98%, and a 600-second measurement time. The application note states that GC workflows typically require 45-60 minutes per sample, while MRR achieves equivalent ppm-level sensitivity in less than 15 minutes, representing roughly a 75% time reduction in this use case.

Figure: MRR is 75% faster compared to GC-MS at identifying and quantitating EG and DEG in PEG-400.
How does MRR help with EtO and acetaldehyde testing in PEG-3350?
In the PEG-3350 application note, MRR is used for targeted testing of ethylene oxide (EtO) and acetaldehyde in a matrix that is notoriously difficult to handle by conventional injection-based workflows. The application note explicitly describes GC-MS as the regulatory mainstay, but highlights the practical challenges of applying it to viscous PEG-3350, including dilution, extra preparation, and careful handling that can reduce throughput and increase variability.
By contrast, the MRR workflow uses headspace sampling to analyze the target impurities directly in prepared PEG-3350 solutions, with no derivatization reported in the method. The application note reports strong linearity for both analytes. It also highlights short acquisition times of about 60 seconds for EtO and 40 seconds for acetaldehyde after headspace equilibration. The application note states that MRR reached LOQs aligned with USP acceptance thresholds for PEG-3350 impurity monitoring, positioning the method as a practical alternative for routine excipient screening.
How does MRR relate to residual solvent analysis?
Residual solvent analysis is another workflow where the difference between GC-MS and MRR is easy to understand. In many laboratories, residual solvents are typically analyzed by headspace GC-MS, often using a single quadrupole detector. The sample is first separated by gas chromatography over an approximately 15-30 minute run, and compounds are then identified and quantified from their mass spectra using calibration standards.
MRR takes a different approach. Instead of relying on chromatographic separation, the sample is analyzed directly and residual solvents are identified and quantified based on their unique MRR spectra. If you want a deeper look at that workflow specifically, explore our Residual Solvent Analysis article.
How does MRR compare to GC-MS for essential oil authenticity and stereoisomer analysis?
One of the clearest differences between GC-MS and MRR appears in structurally similar compounds. Traditional workflows may rely on chromatographic separation, spectral interpretation, or additional confirmatory techniques to distinguish isomers. MRR approaches the problem differently: each analyte produces a distinct rotational spectrum that reflects its three-dimensional structure. This makes MRR especially valuable when molecular formula alone is not enough to confirm identity.
In BrightSpec's natural product application note, compounds such as viridiflorol, globulol, ledol, and epiglobulol are highlighted as a representative challenge. These sesquiterpene alcohols share identical molecular formulas and closely related scaffolds, yet differ in stereochemistry. The application note explains that GC-based methods paired with mass spectral libraries can support broad classification, but may fall short when asked to resolve closely related diastereomers in complex mixtures or when physical reference standards are unavailable or impractical.
Why is MRR useful for stereoisomer identification?
MRR is useful for stereoisomer identification because it encodes three-dimensional molecular structure directly into the measured spectrum. In the essential oil application note, BrightSpec shows that simulated rotational spectra for closely related stereoisomers are distinct and non-overlapping across the same frequency window, even when the molecules share the same formula and similar core structure. That allows analysts to move from a list of plausible candidates to a single, defensible structural assignment without relying on chromatographic separation or physical reference standards.
The application note also positions MRR as a bridge between GC-MS and downstream structural confirmation. Where GC-MS can provide molecular formula and broad classification, and NMR is often reserved for later-stage structural confirmation, MRR shifts structure-level confirmation upstream by using simulated and experimental rotational spectra together. For authenticity testing, chemotype verification, and quality control in natural product workflows, that can be a meaningful operational advantage.
Can MRR replace GC-MS completely?
MRR is not a universal replacement for GC-MS, but is best understood as a highly specific targeted workflow for applications where structural specificity, simplified setup, or faster method readiness matter most. In some laboratories, MRR may serve as a direct alternative for defined assays. In others, it may function as an orthogonal tool alongside GC-MS to strengthen confidence in compound identity, especially when isomers, stereoisomers, or low-level targeted impurities are involved.
- Use GC-MS when: You need broad untargeted screening or a deeply established legacy workflow.
- Use MRR when: You need targeted, structure-specific confirmation with less chromatography-dependent method development.
- Use both together when: You want an orthogonal analytical strategy for difficult or high-consequence decisions.
What are the biggest workflow advantages of MRR over GC-MS?
The biggest workflow advantages of MRR over GC-MS are reduced chromatography-dependent method development, simplified handling for challenging matrices, and stronger specificity for structurally similar compounds. Across BrightSpec's app notes, these advantages appear consistently in both excipient testing and essential oil authenticity use cases.
- Less chromatography-driven method development: Targeted MRR workflows can reduce dependence on retention-based optimization.
- Faster method readiness: Less emphasis on chromatographic tuning before routine analysis.
- High specificity for known targets: Rotational fingerprints can distinguish compounds that are difficult to separate or confirm confidently by conventional means.
- Better fit for difficult matrices: Headspace-based workflows can reduce handling burden in viscous excipient applications.
- Clear path to automation: BrightSpec application notes describe autosampler workflows, queue setup, and batch-friendly operation.
When should pharmaceutical and natural product labs consider MRR?
Pharmaceutical and natural product labs should consider MRR when GC-MS methods are technically valid but operationally inefficient, or when structure-specific confidence matters more than broad untargeted coverage. That includes excipient testing for regulated impurities, volatile analyte screening in difficult matrices, and authenticity workflows where stereochemical misassignment can affect product quality, supplier qualification, or downstream decisions.
For many labs, the most practical way to evaluate MRR is not as a theoretical replacement for every GC-MS method, but as a targeted solution for the assays that create the most friction. If a workflow is slowed by matrix-specific preparation, chromatography-heavy method development, or uncertainty around closely related compounds, MRR may be worth evaluating as a simpler and more specific alternative.
Frequently Asked Questions
What is the difference between GC-MS and MRR?
GC-MS separates compounds chromatographically before mass analysis, while MRR identifies target molecules by their unique rotational spectra. For targeted analyses, MRR can reduce chromatography-dependent method development while improving specificity for structurally similar compounds.
Can MRR replace GC-MS for excipient testing?
In targeted excipient testing workflows, MRR can be a strong alternative to GC-MS. BrightSpec application notes show direct quantitation of ethylene glycol, diethylene glycol, ethylene oxide, acetaldehyde, and formaldehyde in pharmaceutical excipient matrices with simplified workflows and specific detection.
How does MRR compare with GC-MS for residual solvent analysis?
Residual solvent analysis is commonly performed by headspace GC-MS, where compounds are separated chromatographically and then identified from their mass spectra. MRR uses a different workflow: samples are analyzed directly and residual solvents are identified and quantified from their unique MRR spectra. That makes residual solvent analysis a relevant example of how MRR can simplify targeted workflows compared with conventional GC-MS.
Why is MRR useful for essential oil authenticity testing?
MRR is especially useful for essential oil authenticity because it can distinguish structurally similar compounds, including stereoisomers, without relying on chromatographic separation. This makes it valuable for confirming composition and detecting authenticity issues in complex natural product mixtures.
Does MRR require chromatographic separation for these targeted assays?
No. In the targeted workflows described here, MRR does not rely on chromatographic separation to confirm analytes. That can simplify routine testing compared with GC-MS methods that depend on retention-based separation and interpretation.
Where can I learn more about BrightSpec MRR application notes?
Explore BrightSpec application notes covering excipient testing and essential oil authenticity to see workflow comparisons, analytical performance, and real examples of MRR used for targeted chemical analysis.
Conclusion: Is MRR a practical alternative to GC-MS?
For the right applications, yes. BrightSpec's application notes show that MRR can be a practical alternative to GC-MS when laboratories need targeted, structure-specific analysis with less chromatography-dependent method development and a simpler operational path. In excipient testing, MRR supports direct, headspace-based workflows for compounds such as ethylene glycol, diethylene glycol, ethylene oxide, and acetaldehyde. In essential oil and natural product analysis, it provides a structure-level approach for distinguishing closely related stereoisomers that can challenge conventional workflows.
Rather than replacing every GC-MS method, MRR is best viewed as a modern targeted analytical tool: one that can simplify selected workflows, strengthen confidence in difficult identifications, and help laboratories move faster when the cost of ambiguity is high.
Want to see the workflows in detail? Explore BrightSpec's application notes to compare GC-MS and MRR across excipient testing and essential oil authenticity use cases.